(Part 23 of Dr. Pagtakhan’s column on Medisina at Politika.)
January 16, 2021- Canada’s first recipients of the Pfizer-BioNTech’s COVID-19 vaccine –the 89-year-old Gisele Levesque (a resident at the Saint-Antoine long-term care home in Quebec City) and Anita Quidangen (a personal support worker at the multilingual, non-profit nursing home Rekai Center in Toronto, Ontario) - got their first shots of hope on Monday December 14, 2020 at 11:25 a.m. and 12:03 p.m., respectively, one day after the first vaccine supply arrived. Barely a month since on the 13th of this month, more than half a million doses (532,050) of this and subsequent additional vaccine by Moderna have been delivered to all provinces and territories and 73% (388,623 of the doses) have been administered. What, indeed, has been the progress on Canada’s COVID-19 mass vaccination drive?
First, a Quick Reminder
How to Protect Yourself (and Others) When Going Out:
- Wear a mask that covers your nose and mouth.
- Stay 6 feet apart from others who don’t live with you.
- Avoid crowds. The more people you are in contact with, the more likely you are exposed.
- Avoid poorly ventilated indoor spaces.
- Wash your hands often with soap and water. Use hand sanitizer if soap and water aren’t available.
In fact, the above-noted first three action-measures are also the 3 important ways to slow the spread of COVID-19. Thus, protecting yourself and others helps slow down the spread of contagion.
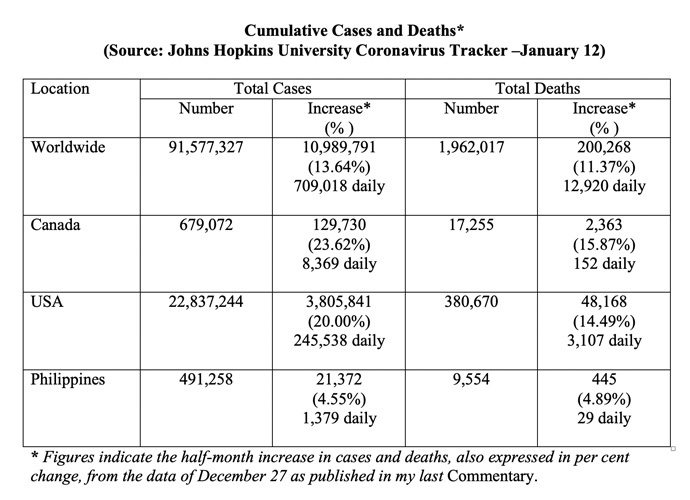
Current Pandemic Situation (Table below)
Globally, the half-month increase in the total diagnosed came to over 10 million cases, that is, an average of over 700 thousand cases daily; the total cumulative cases have surpassed the 91 million mark. Deaths increased by 11.37% - a total daily average of over 12,000.
Compared to the global figures, Canada showed a half-month increase of over 129,000 cases, that is, a total daily average of over 8,000. Deaths increased by over 15% - a daily total average of over 150.
For the U.S.A., the half-month increases expressed in per cent in both cases diagnosed and deaths seen were similar to that of Canada in the magnitude of upward trends.
The increases in cases and deaths for the Philippines over the same half-month period were of the same magnitude compared to its preceding half-month period.
Overall, the pandemic figures remain alarming. In Ontario, Premier Dough Ford has declared its second state of emergency effective January 14; it will be in effect for at least 28 days. Thus, due attention has been given to the mass vaccination program.
Supply and Distribution of Vaccines
Canada’s recent procurement from Pfizer of another 20 million COVID-19 vaccine doses, as announced by Prime Minister Justin Trudeau on January 12 (Ryan Patrick Jones: CBC News, Jan 12, 2021) brings the total number of doses (of the two vaccine brands) scheduled to arrive this year to 80 million. Earlier, Procurement Minister Anita Anand announced no further order was confirmed with Moderna since it cannot guarantee the arrival of supply by September.
Since the vaccine is given in two doses per person, the country is assured of supply for 40 million Canadians. Canada’s population in 2020 from age 18 years and older is 30.76 million. The population target for the Pfizer-BioNTech vaccine starts at 16 years of age (18 years for Moderna vaccine). Only for the Pfizer-BioNTech vaccine, “adolescents 12 to 15 years of age may be offered (1) if a risk assessment deems that the benefits of vaccination outweigh the potential risks for the individual and (2) if informed consent includes discussion about the insufficient evidence in these populations.” I estimate only a small percentage of vaccines would come from this population sub-group.
Hence, Canadians can rest assured they will have more than adequate supply to fully vaccinate our total eligible population and thereby reach the desired population immunity before the end of this year. Said the Prime Minister: "From our agreements with Pfizer and Moderna alone ... we are on track to have every Canadian who wants a vaccine to receive one by September (this year)."
As of January 13th, 532,050 doses (joint Pfizer/BioNTech and Moderna vaccines) have been distributed to all provinces and territories as shown on the Public Health Agency Canada website. Some 73.00% (388,623) have been administered. The distribution chart is updated weekly “to allow them to plan and manage their rollout processes. “The numbers are charted for weekly distribution until the end of next month. Minister Anand estimates 6 million more doses by March and another 20 million between April and June — enough to vaccinate 13 million people during this period, that is, nearly half (42%) of the eligible community. This would help ease the concern of some provinces that have been able to have their doses administered faster than the delivery of supply, as experienced by Alberta and British Columbia.
Speed of Vaccine Distribution and Actual Vaccination
The continuing surge in cases and deaths following the holiday season and the identification of new and more readily transmissible virus strains have created greater urgency for speed of vaccination to protect more of the population. Taking into account this epidemiologic situation, the feasibility of varying the time intervals between the two doses as earlier set by both Pfizer-BioNTech and Moderna have been raised. The B.C. Centres for Disease Control, the National Advisory Committee on Immunization (NACI), and international research groups said recently - with guidance from the World Health Organization - there is no evidence that a longer wait before administering the second dose affects immunity. Looking at this issue, the Public Health Agency of Canada has noted uncertainties around the impact of extending the intervals between the first and second dose, including the uncertainties that could “negatively impact public trust in the COVID-19 immunization program, the COVID-19 response, and vaccines in general; perpetuate perceptions that certain populations are exposed to an experimental approach; increase vaccine hesitancy for COVID-19 vaccines and vaccines in general, especially if individuals vaccinated with one dose get disease due to insufficient protection.” In light of these observations, I feel it is best not to pursue the option of lengthening the intervals but, instead, focus work on the logistics and certainty of delivery schedules.
Managing the Vaccine Queue
Globe and Mail health columnist Andre Picard (GM: Jan 11, 2021) has made the following observations on the ethics of the vaccination drive with respect to managing the queue for the short supply of the vaccines:
- “Recognize that there are non-debatable priority groups for the initial supply:
- elderly Canadians in institutional settings and front-line health workers in direct contact with COVID-19 patients; and
- people in high-risk settings such as group homes, prisons and remote Indigenous communities;
- Recognize that as more supply becomes available, we go out into the community and target:
- elders who live on their own as well as their personal community health workers;
- people living with disabilities;
- essential workers - grocery store clerks, food/hospitality workers, teachers; and
- more vulnerable community members, beginning with those experiencing homelessness;
- Recognize that after the above-noted groups, prioritization becomes more difficult; and
- Recognize that most of us will be waiting months for a vaccine; that not all hospital staff should have first dibs; that cronyism cannot be tolerated and neither can favouritism.”
Indeed, reports have come out that some staff from a hospital setting jumped the queue presumably on the basis that appointments from the prioritized groups failed to keep their schedule and the prepared vials could be wasted. While it is critical not to waste vaccines, particularly from already open vials, the answer calls for more careful planning.
Adverse Reactions
Health Canada has launched a site from tracking vaccine safety. It looks at (1) whether the side effects documented are more severe, or greater in number, than what the trials saw; and (2) whether anything has happened that was not observed during the clinical trials. To date, the numbers encountered are in line with those seen during the clinical trials. No unexpected side effect has been reported.
Rey D. Pagtakhan, P.C., O.M., LL.D., Sc.D., M.Sc., M.D. – former cabinet minister and Parliamentary Secretary to Canada’s Prime Minister and retired lung specialist and professor – graduated from the University of the Philippines. He did postgraduate studies/training at Washington University and University of Manitoba and spent a sabbatical year as Visiting Professor at the University of Arizona. Author/co-author of chapters and papers for medical textbooks and journals, he writes the column Medisina at Politika for two Canadian Filipino community publications. Widely lectured, he spoke on the “Global Threat of Infectious Diseases” at the G-8 Science Ministers and Advisors Carnegie Group Meeting held June 13-15, 2003 in Berlin, Germany.














